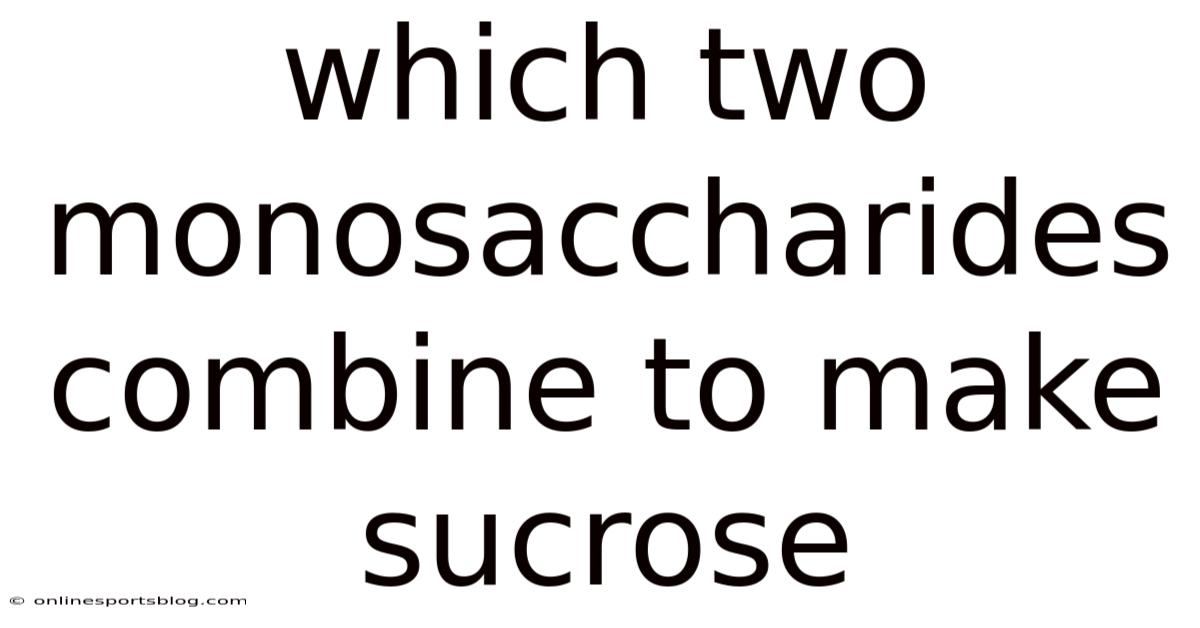The nuanced dance of biochemical reactions underpins the very fabric of life on Earth, weaving together the smallest units of sugar into monumental molecules that sustain ecosystems, nourish organisms, and shape cultural traditions. Worth adding: as researchers continue to unravel the mechanisms behind such transformations, the study of sucrose formation remains a cornerstone of modern biochemistry, bridging the gap between abstract concepts and tangible reality. Yet sucrose’s existence is a testament to the precision of natural chemistry, arising from the union of two distinct monosaccharides—glucose and fructose—each contributing a unique profile of carbon, hydrogen, and oxygen atoms. And understanding this formation process not only illuminates the molecular basis of sucrose but also underscores the broader implications of biochemical synergy in sustaining life. Among these important molecules lies sucrose, a disaccharide renowned for its role as a universal sweetener and energy source. This symbiotic relationship between glucose and fructose, though seemingly simple on the surface, reveals a complexity that has fascinated scientists for centuries. The interplay of these components exemplifies how fundamental processes can evolve into widespread applications, influencing everything from agriculture to medicine, thereby highlighting sucrose’s enduring significance in both natural and industrial contexts.
Counterintuitive, but true.
Understanding the Components
At the heart of sucrose’s composition lies glucose, a six-carbon sugar polymer derived from glucose units linked through glycosidic bonds, while fructose presents as a four-carbon structure with a different spatial arrangement of atoms. These two monosaccharides, though structurally distinct, share a commonality that enables their combination: the shared ability to undergo hydrolysis under specific conditions. Hydrolysis, the process of breaking down disaccharides into monosaccharides, is a cornerstone of carbohydrate metabolism, serving as a critical step in energy extraction and nutrient absorption. Still, sucrose’s unique properties stem not merely from the mere union of these two molecules but from their complementary chemical properties. Glucose, with its straight-chain configuration, readily participates in metabolic pathways, whereas fructose, often metabolized differently in the liver, contributes to its role as a preferred sweetener in human consumption. This duality positions glucose and fructose as key players in the biochemical landscape, yet their partnership in sucrose formation introduces a layer of specificity that distinguishes it from other disaccharides. The precision required to balance their reactivity ensures that sucrose remains a stable yet versatile compound, capable of serving diverse functions across biological systems and human applications alike Small thing, real impact. That alone is useful..
The Chemical Process
The transformation from glucose and fructose to sucrose occurs through a process termed hydrolysis, which is catalyzed by enzymes such as sucrose hydrolase or human digestive enzymes like sucrase. This enzymatic activity breaks the glycosidic bond linking glucose and fructose, releasing individual monomers that can then interconvert into sucrose. Still, the formation of sucrose is not merely a passive reaction; it is a delicate equilibrium that depends on temperature, pH, and the presence of specific ions. In biological contexts, this equilibrium is tightly regulated to prevent degradation or improper utilization of the resulting sugar. Conversely, in industrial settings, controlled conditions are applied to ensure efficient conversion, maximizing yield and minimizing waste. The chemistry involved here is further complicated by the presence of water molecules and the role of ATP in energy transfer during metabolic processes. Additionally, the molecular geometry of glucose and fructose matters a lot, as their different conformations influence how readily they can interact with each other. This interplay between structure and function underscores why sucrose’s formation is both a biological necessity and a subject of scientific inquiry. Understanding these nuances allows scientists to manipulate the process for practical purposes, whether in food production, pharmaceutical formulations, or energy storage solutions.
Historical Context and Cultural Significance
Historically, sucrose has held a central role in human history, serving as a cornerstone of trade and cultural exchange. Its discovery in ancient civilizations, particularly in Egypt and India, revolutionized culinary practices by enabling the creation of sweetened beverages, preserved foods, and desserts that would otherwise have been impractical. The monomers glucose and fructose, derived from sugar cane and sugar beet, became staples in global diets, their combination allowing for the production of a sweetener that was both potent and versatile. Yet beyond its practical applications, sucrose carries symbolic weight, often associated with luxury, celebration, and shared heritage. In many cultures, the act of breaking down sucrose into its components is celebrated as a metaphor for the dissolution of barriers or the reconciliation of disparate elements into something harmonious. This duality—practical utility paired with cultural resonance—makes sucrose more than just a chemical compound; it becomes a narrative embedded within human history. The persistence of sucrose in modern cuisine
The persistence of sucrose in moderncuisine reflects its adaptability and enduring utility. Its ability to dissolve readily, its role in caramelization for flavor development, and its compatibility with other ingredients make it a preferred choice in baking, confectionery, and beverage industries. Practically speaking, in pharmaceuticals, sucrose serves as an excipient in drug formulations, leveraging its stability and bioavailability. On top of that, while health-conscious trends have prompted a shift toward low-calorie alternatives, sucrose remains indispensable in many applications due to its unique properties. Worth adding, advancements in controlled crystallization techniques and microencapsulation have expanded its use in functional foods, where sustained sweetness and texture are critical. Even in energy storage research, scientists explore sucrose-based biofuels, though challenges related to its metabolic impact persist But it adds up..
Culturally, sucrose continues to symbolize celebration and connection. Modern festivals, from Diwali sweets to Christmas cakes, often center around sucrose-laden treats, reinforcing its role as a marker of joy and communal identity. In art and media, sucrose frequently appears as a metaphor for sweetness in life or the complexities of excess, reflecting societal attitudes toward consumption. Its historical journey from ancient trade routes to global ubiquity underscores a narrative of human innovation and cultural adaptation.
Pulling it all together, sucrose embodies a remarkable interplay of
To wrap this up, sucrose embodiesa remarkable interplay of scientific ingenuity and cultural legacy, transcending its chemical identity to become a symbol of human ingenuity and shared experiences. Its journey from ancient kitchens to modern laboratories and celebrations reflects humanity’s ability to adapt and innovate while preserving traditions. As both a functional ingredient and a metaphor for sweetness in life, sucrose continues to bridge past and present, reminding us that even the most ordinary compounds can carry profound narratives. In an era of rapid technological and dietary change, its enduring presence challenges us to reconsider how we value not just what we consume, but the stories embedded within it. When all is said and done, sucrose is more than a sweetener—it is a testament to the layered dance between necessity and symbolism in shaping our world Which is the point..
Yet, this very ubiquity masks complex geopolitical and ethical dimensions. Here's the thing — the global sucrose economy remains deeply entangled with historical patterns of colonization and contemporary labor challenges, particularly in regions where sugarcane is a primary crop. Questions of fair trade, environmental degradation from monoculture farming, and water usage cast a shadow on its sweet narrative, reminding us that the commodity’s journey from field to table is rarely neutral. Adding to this, as climate change threatens traditional growing regions, the future stability of sucrose supply chains prompts a reevaluation of agricultural resilience and diversification Simple, but easy to overlook. Turns out it matters..
These layered realities—scientific marvel, cultural keystone, economic linchpin, and ethical challenge—prevent sucrose from being merely a passive ingredient. It is an active participant in global systems, a substance that forces conversations about sustainability, equity, and the true cost of sweetness. Its story is not one of simple pleasure, but of detailed human systems, where desire, necessity, and consequence are inextricably blended.
All in all, sucrose embodies a remarkable interplay of scientific ingenuity and cultural legacy, transcending its chemical identity to become a symbol of human ingenuity and shared experiences. Its journey from ancient kitchens to modern laboratories and celebrations reflects humanity’s ability to adapt and innovate while preserving traditions. As both a functional ingredient and a metaphor for sweetness in life, sucrose continues to bridge past and present, reminding us that even the most ordinary compounds can carry profound narratives. In an era of rapid technological and dietary change, its enduring presence challenges us to reconsider how we value not just what we consume, but the stories embedded within it. At the end of the day, sucrose is more than a sweetener—it is a testament to the detailed dance between necessity and symbolism in shaping our world Worth keeping that in mind..
Some disagree here. Fair enough.