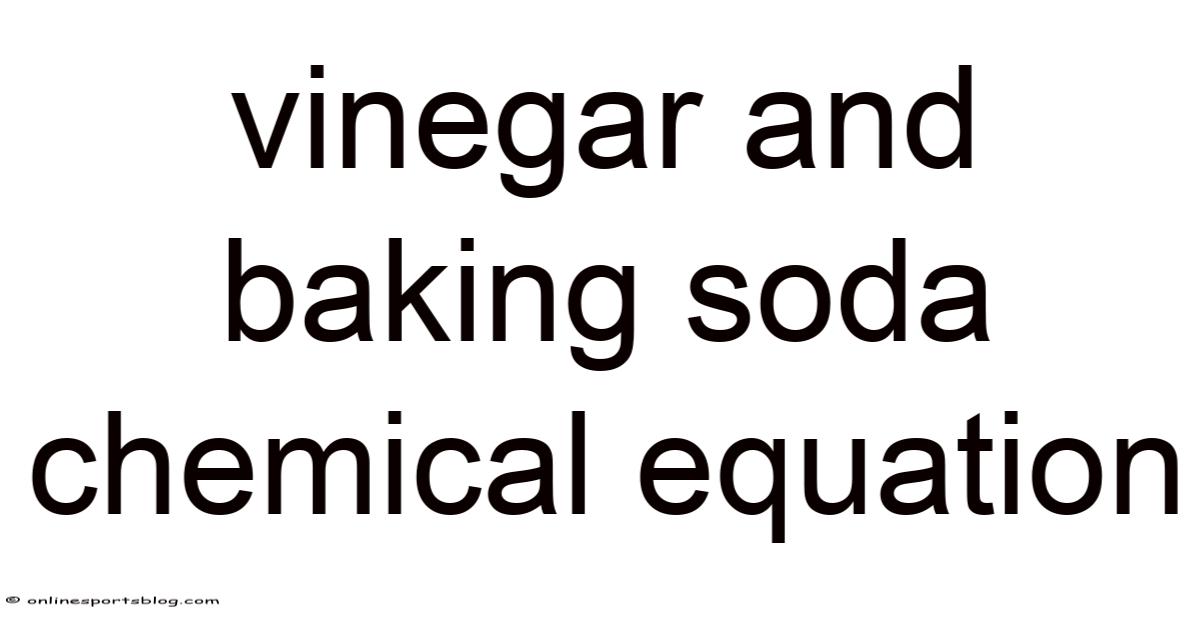Understanding the chemistry behind vinegar and baking soda can open up a world of possibilities in the kitchen. These two simple ingredients are not just household staples; they play a crucial role in various reactions that enhance flavor, texture, and even the science of baking. But in this article, we will explore the chemical equation that forms when vinegar and baking soda meet, dig into its practical applications, and uncover why this reaction is so powerful. Whether you're a student, a home cook, or a science enthusiast, this guide will provide you with a clear and engaging understanding of this fascinating interaction Easy to understand, harder to ignore..
The interaction between vinegar and baking soda is a classic example of a chemical reaction that produces carbon dioxide gas. So this process is not only visually exciting but also scientifically significant. By breaking down the reaction, we can see how these two substances combine to create a fizzy effect that many people enjoy. So this reaction is a key part of what makes certain recipes rise and what gives certain foods their signature texture. Understanding this process helps us appreciate the science behind everyday cooking The details matter here..
To begin with, let’s clarify what vinegar and baking soda are. That said, Vinegar is a common household liquid made primarily of acetic acid, which is a weak acid. It comes in various forms, such as white vinegar, apple cider vinegar, or even homemade versions. Because of that, on the other hand, baking soda, also known as sodium bicarbonate, is a versatile substance often used in baking and cleaning. Plus, it is a mild base that reacts with acids to produce bubbles and lift dough. When these two substances come into contact, something magical happens.
The chemical equation that describes this interaction is quite straightforward. When vinegar, which contains acetic acid, meets baking soda, the following reaction takes place:
Acetic acid + Sodium bicarbonate → Carbon dioxide + Water + Sodium acetate
This equation is essential for understanding how the reaction works. Still, let’s break it down further. Acetic acid, the main component of vinegar, reacts with sodium bicarbonate, the key ingredient in baking soda. The result is the release of carbon dioxide gas, which creates the bubbly effect we see in many recipes. Additionally, the reaction produces water and a salt called sodium acetate. This process not only adds volume but also improves the texture of baked goods And that's really what it comes down to..
The importance of this reaction extends beyond the kitchen. By observing how vinegar and baking soda interact, learners can better understand the principles of chemical change. It is a fundamental concept in chemistry that helps students grasp the basics of acid-base reactions. This knowledge is valuable not just in the classroom but also in real-world applications.
This changes depending on context. Keep that in mind.
Now, let’s explore the practical implications of this reaction. This is because the carbon dioxide produced during the reaction expands as it escapes, helping the dough rise. In baking, the interaction between vinegar and baking soda is essential for leavening. When a recipe calls for baking soda, adding a splash of vinegar can help create a light and fluffy texture in cakes and cookies. Many recipes use this method to achieve the perfect consistency in their baked goods.
In addition to baking, this reaction is used in various cleaning products. And for instance, a common household cleaner uses a mixture of vinegar and baking soda to tackle tough stains and odors. The reaction helps break down grime and neutralize odors, making it an effective and eco-friendly cleaning solution. This dual-purpose use highlights the versatility of vinegar and baking soda in both culinary and domestic settings That's the part that actually makes a difference..
The scientific explanation of this reaction is rooted in the properties of acetic acid and sodium bicarbonate. Acetic acid is a weak acid that can donate protons, while sodium bicarbonate is a base that can accept protons. When these two substances mix, a chemical equilibrium is established. The reaction shifts to produce more products, resulting in the formation of carbon dioxide gas. This process is known as a neutralization reaction, where an acid and a base combine to form a salt and water.
You'll probably want to bookmark this section.
Understanding this reaction also helps in explaining why certain foods require baking soda for proper rising. To give you an idea, in bread making, the sodium bicarbonate in baking soda reacts with the acids in the dough, creating bubbles that help the bread expand. This is why bakers often use this method to achieve a light and airy texture.
Worth pointing out that while vinegar and baking soda are safe to use together, they can also produce harmful fumes if not handled properly. When these substances are combined, they release carbon dioxide gas, which can create a pressure build-up. This is why it is advisable to perform this reaction in a well-ventilated area or with a spray bottle to control the release of gas.
For those interested in experimenting with this reaction, there are several fun and creative ways to apply it. Practically speaking, you can try making homemade bread by incorporating vinegar and baking soda into your dough. Alternatively, you could use this reaction to clean surfaces by mixing it with water and a few drops of dish soap. The result might surprise you with the texture and rise of your creation. The fizzing action helps lift dirt and grime away from your kitchen tools Not complicated — just consistent. Turns out it matters..
The FAQs section is essential for addressing common questions that readers might have. Here's a good example: many people wonder about the safety of using vinegar and baking soda together. It is important to avoid inhaling the fumes and to confirm that the mixture is well diluted before use. Also, the answer is generally safe, as long as proper precautions are taken. Additionally, some individuals may have sensitivities to certain ingredients, so it is always a good idea to test a small amount first That alone is useful..
Another question that often arises is about the effectiveness of this reaction in different recipes. Practically speaking, for example, using too much vinegar might lead to an overly acidic flavor, while too much baking soda could cause the mixture to fizz excessively. While vinegar and baking soda can work well in many recipes, the outcome depends on the proportions used. The answer lies in the balance of ingredients. Because of this, understanding the ratio is crucial for achieving the desired results Not complicated — just consistent..
Some disagree here. Fair enough.
In the realm of education, this topic serves as an excellent teaching tool. Which means by analyzing the chemical equation and its effects, learners can develop critical thinking skills and a deeper understanding of chemistry. So it allows students to connect theoretical concepts with practical applications. This approach not only enhances their knowledge but also fosters a sense of curiosity about the world around them.
Honestly, this part trips people up more than it should.
Beyond that, the conclusion of this article highlights the significance of vinegar and baking soda in our daily lives. Beyond their culinary uses, these ingredients demonstrate the beauty of chemistry in action. Day to day, whether you're a beginner or an expert, understanding this reaction empowers you to experiment and innovate in the kitchen. It encourages you to see the science behind what you cook and eat, making your culinary adventures more meaningful Simple as that..
Boiling it down, the chemical equation between vinegar and baking soda is more than just a simple reaction. It is a gateway to understanding the science of cooking, a tool for enhancing textures, and a reminder of the importance of chemistry in everyday life. By embracing this knowledge, you not only improve your baking skills but also deepen your appreciation for the processes that shape our meals. This article has provided a comprehensive overview of this fascinating interaction, ensuring that you leave with a clearer understanding of its importance.