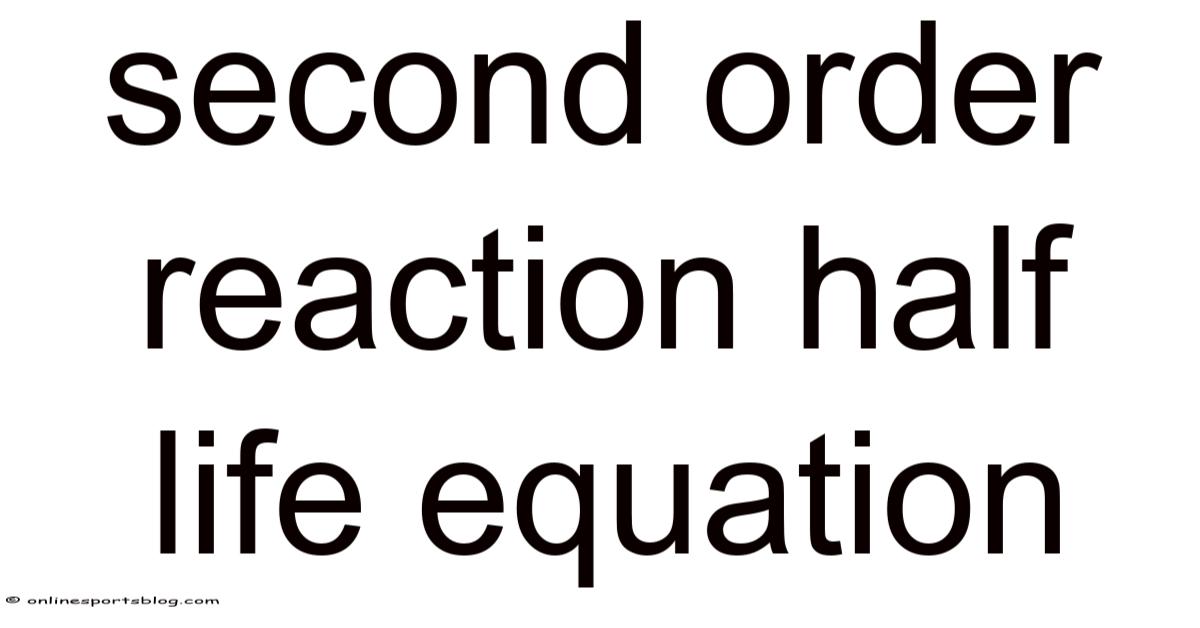Second Order Reaction Half Life Equation
Understanding how the concentration of reactants changes over time is one of the most fundamental concepts in chemical kinetics. In practice, the second order reaction half life equation tells us that the time it takes for a reactant to reduce to half its initial concentration depends on that initial concentration itself. Among the different reaction orders, second order reactions hold a special place because their half-life behavior differs significantly from first order reactions. This simple yet powerful relationship reveals a lot about the mechanism and dynamics of a chemical reaction That's the whole idea..
Worth pausing on this one.
What Is a Second Order Reaction?
A second order reaction is a reaction where the rate of reaction is proportional to the product of the concentrations of two reactant molecules, or to the square of the concentration of a single reactant. The general rate law can be written as:
Rate = k[A]² or Rate = k[A][B]
Where k is the rate constant and [A] or [B] represents the molar concentration of the reactants. On top of that, in a second order reaction with a single reactant, doubling the initial concentration will quadruple the initial rate of the reaction. This distinguishes it clearly from first order reactions, where the rate is directly proportional to the concentration of one reactant.
The official docs gloss over this. That's a mistake.
Common examples of second order reactions include the decomposition of nitrogen dioxide (2NO₂ → 2NO + O₂) and the reaction between nitric oxide and hydrogen gas (2NO + 2H₂ → N₂ + 2H₂O) Still holds up..
Deriving the Second Order Half Life Equation
To understand the half life equation, we start with the integrated rate law for a second order reaction involving a single reactant:
1/[A] = 1/[A]₀ + kt
Where [A] is the concentration at time t, [A]₀ is the initial concentration, and k is the rate constant. This equation is derived by separating variables and integrating the rate law d[A]/dt = -k[A]² from [A]₀ to [A] over time 0 to t.
Now, the half-life, denoted as t½, is defined as the time required for the concentration of the reactant to drop to half of its initial value. So at t = t½, we have:
[A] = [A]₀ / 2
Substituting this into the integrated rate law:
1/([A]₀/2) = 1/[A]₀ + k·t½
Simplifying the left side:
2/[A]₀ = 1/[A]₀ + k·t½
Subtracting 1/[A]₀ from both sides:
1/[A]₀ = k·t½
That's why, the second order half life equation is:
t½ = 1 / (k[A]₀)
This equation shows that the half-life of a second order reaction is inversely proportional to the initial concentration of the reactant. Unlike first order reactions, where the half-life is constant regardless of concentration, the half-life in a second order reaction changes as the reaction progresses Easy to understand, harder to ignore..
Key Differences Between First and Second Order Half Lives
How second order half lives compare to first order half lives stands out as a key concepts to grasp. In a first order reaction, the half-life equation is:
t½ = 0.693 / k
This value remains constant throughout the entire reaction, no matter what the starting concentration is. The half-life is independent of [A]₀.
In contrast, for a second order reaction:
t½ = 1 / (k[A]₀)
Here, the half-life decreases as the initial concentration increases. If you start with a higher concentration, the reactant will reach half its value more quickly. This is a direct consequence of the rate law, where the reaction rate depends on the square of the concentration.
This difference is not just a mathematical curiosity. It has practical implications in fields like pharmacokinetics, atmospheric chemistry, and enzyme kinetics, where knowing how quickly a substance degrades or reacts is essential Not complicated — just consistent. But it adds up..
Graphical Representation
The behavior of a second order reaction can also be understood through graphs. That's why if you plot 1/[A] versus time t, you get a straight line with a slope equal to k and a y-intercept equal to 1/[A]₀. This is because the integrated rate law is linear in this form Simple, but easy to overlook..
Looking at it differently, if you plot [A] versus time for a second order reaction, the curve is steeper at the beginning and gradually levels off, unlike the exponential decay seen in first order reactions. The half-life points on this curve become progressively shorter as the reaction proceeds, reflecting the inverse relationship with concentration.
Plotting t½ against 1/[A]₀ also yields a straight line, confirming the relationship t½ = 1/(k[A]₀) where the slope equals 1/k.
Worked Example
Let us say a second order reaction has a rate constant k = 0.02 L·mol⁻¹·s⁻¹ and the initial concentration of the reactant is 0.10 mol/L. What is the half-life?
Using the equation:
t½ = 1 / (k[A]₀)
t½ = 1 / (0.02 × 0.10)
t½ = 1 / 0.002
t½ = 500 seconds
Now, if the initial concentration is doubled to 0.20 mol/L, the new half-life becomes:
t½ = 1 / (0.02 × 0.20) = 1 / 0.004 = 250 seconds
As expected, doubling the concentration halves the half-life, confirming the inverse proportionality.
Why Does This Matter in Real Life?
The second order reaction half life equation is not just a theoretical exercise. It appears in numerous real-world scenarios. Even so, in atmospheric chemistry, reactions between pollutants often follow second order kinetics, and understanding their half-lives helps model how quickly harmful substances degrade. In biochemistry, many enzyme-catalyzed reactions show second order behavior when the substrate concentration is low, and knowing the half-life aids in designing drug dosing schedules.
Not obvious, but once you see it — you'll see it everywhere.
Even in everyday contexts, the principle behind the half-life equation reminds us that the speed of a reaction is not always constant. It can depend on how much reactant is present at the start, and recognizing this can change how we interpret experimental data.
Frequently Asked Questions
Does the half-life of a second order reaction ever become constant? No. For a simple second order reaction with a single reactant, the half-life continuously decreases as the concentration drops. Only first order reactions have a constant half-life.
Can the second order half life equation apply to reactions with two different reactants? The general integrated rate law for a second order reaction of the type A + B → products is more complex. The simple half life equation t½ = 1/(k[A]₀) applies specifically when the initial concentrations of both reactants are equal or when the reaction involves a single reactant That alone is useful..
How do you determine if a reaction is second order experimentally? You can determine the reaction order by plotting different functions of concentration against time. If a plot of 1/[A] versus time gives a straight line, the reaction is second order. The slope of that line equals the rate constant k.
What happens to the half-life if the rate constant changes? Since t½ = 1/(k[A]₀), increasing the rate constant k will decrease the half-life. A larger k means the reaction proceeds faster, so it takes less time to reach the half concentration point.
Conclusion
The second order reaction half life equation, t½ = 1 / (k[A]₀), is a cornerstone of chemical kinetics. It captures the essential relationship that in second order reactions, the time to halve the reactant concentration depends on how much reactant you start with. Understanding this equation, along with its derivation and graphical implications, equips students and professionals alike to analyze reaction data, predict reaction behavior, and apply kinetic principles to real-world problems And it works..
Whether you are modeling atmospheric pollutants, optimizing industrial processes, or studying enzyme kinetics in the lab, this equation serves as a fundamental tool in your kinetic toolkit And that's really what it comes down to..
It is worth remembering that the beauty of chemical kinetics lies in its predictive power. The half-life equation does not merely describe what has already happened in a reaction—it allows us to anticipate what will happen under different conditions. By knowing the initial concentration and the rate constant, we can forecast how long a system will take to reach any given point of completion, not just the halfway point That alone is useful..
As modern analytical techniques continue to advance, our ability to measure rate constants with greater precision improves, making these equations even more valuable in both research and industrial applications. From designing more efficient catalytic converters to developing better pharmaceutical formulations, the principles embedded in the second order half-life equation remain relevant and essential That's the whole idea..
To keep it short, while the equation t½ = 1/(k[A]₀) may appear simple at first glance, it encapsulates a profound truth about the nature of second order reactions: their progress is inherently linked to the concentration of reactants present. This dependence distinguishes them from first order reactions and underscores the importance of context in chemical kinetics. By mastering this relationship, chemists gain deeper insight into reaction dynamics and the ability to harness them for practical ends No workaround needed..