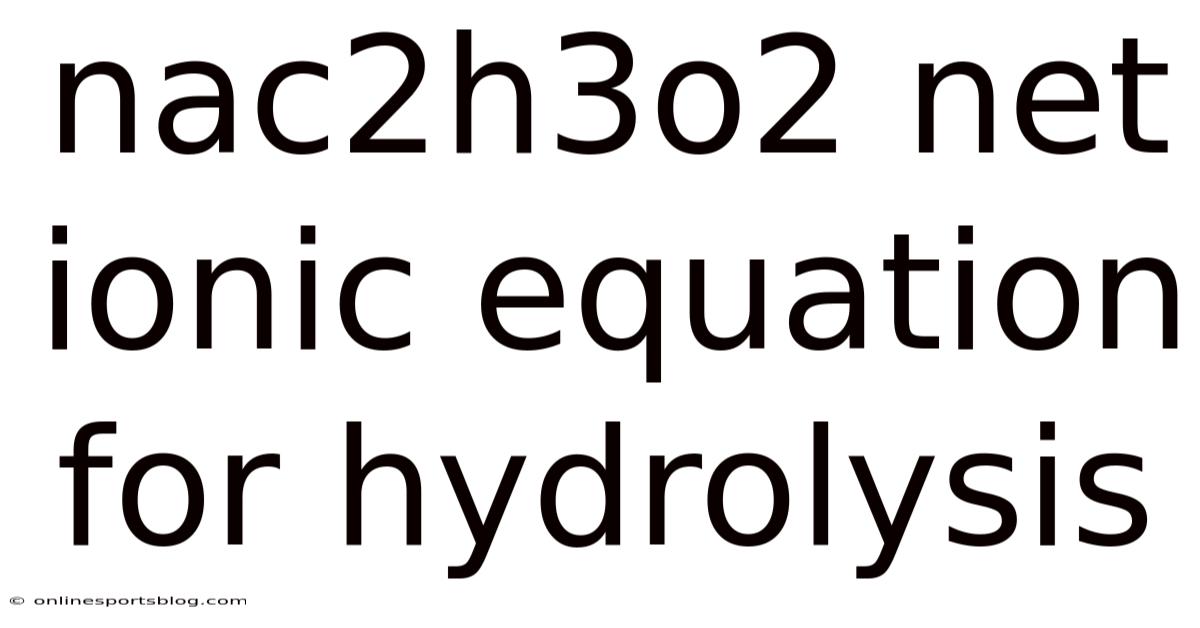The Net Ionic Equation for the Hydrolysis of NaC₂H₃O₂
When sodium acetate (NaC₂H₃O₂) dissolves in water, the resulting solution is not neutral; it is distinctly basic. This fundamental observation is a direct consequence of hydrolysis, a specific type of reaction where an ion from a salt interacts with water molecules. Understanding the precise net ionic equation for hydrolysis of sodium acetate is crucial for predicting pH, explaining buffer systems, and mastering acid-base chemistry. This article will deconstruct the process, moving from the salt’s dissociation to the final, simplified net ionic equation that captures the essential chemical change.
Understanding the Players: Sodium Acetate and Its Ions
Sodium acetate is the salt formed from a strong base, sodium hydroxide (NaOH), and a weak acid, acetic acid (CH₃COOH). This parentage is the key to its hydrolytic behavior. When solid NaC₂H₃O₂ is introduced into water, it undergoes complete dissociation because it is a soluble salt of a Group 1 metal.
The dissociation equation is: NaC₂H₃O₂(s) → Na⁺(aq) + C₂H₃O₂⁻(aq) (Often written as CH₃COO⁻ for the acetate ion)
This process yields two aqueous ions: the sodium cation (Na⁺) and the acetate anion (CH₃COO⁻). The sodium ion is the conjugate acid of the strong base NaOH. As such, it is an extremely weak acid and does not react with water to any appreciable extent. It is a spectator ion. The acetate ion, however, is the conjugate base of the weak acid acetic acid. It possesses a significant affinity for protons (H⁺), making it reactive toward water. This reactivity is the heart of hydrolysis.
The Hydrolysis Reaction: Acetate Ion Attacks Water
Hydrolysis, in this context, refers specifically to the reaction of the acetate ion with water. The acetate ion acts as a Bronsted-Lowry base, accepting a proton from a water molecule. This proton transfer produces molecules of acetic acid and hydroxide ions.
The molecular-level reaction is: CH₃COO⁻(aq) + H₂O(l) ⇌ CH₃COOH(aq) + OH⁻(aq)
This is an equilibrium reaction, as indicated by the double arrow. The equilibrium lies heavily to the left because acetic acid (CH₃COOH) is a weak acid, meaning its conjugate base (CH₃COO⁻) is relatively strong, but not strong enough to pull the reaction completely to the right. The production of hydroxide ions (OH⁻) is the direct cause of the solution’s basic pH.
Deriving the Net Ionic Equation: A Step-by-Step Guide
The net ionic equation is the simplified version of a chemical reaction that shows only the species that actually undergo a chemical change. Spectator ions, which appear unchanged on both sides of the equation, are omitted. For the hydrolysis of sodium acetate, we follow a standard procedure:
-
Write the Complete Ionic Equation: Start by dissociating all strong electrolytes (soluble salts, strong acids, strong bases) into their ions. Leave weak electrolytes (weak acids, weak bases, water) and insoluble solids in their molecular forms.
- NaC₂H₃O₂ is a strong electrolyte → Na⁺(aq) + CH₃COO⁻(aq)
- H₂O is a weak electrolyte → H₂O(l) (kept whole)
- CH₃COOH is a weak acid → CH₃COOH(aq) (kept whole)
- NaOH is a strong base, but it is a product here. We consider its dissociation only if it were a reactant. Since OH⁻ is produced, we write it as the ion. The complete ionic equation becomes: Na⁺(aq) + CH₃COO⁻(aq) + H₂O(l) ⇌ CH₃COOH(aq) + Na⁺(aq) + OH⁻(aq)
-
Identify and Cancel Spectator Ions: Spectator ions are those that appear identically on both the reactant and product sides. In this equation, the Na⁺(aq) ion is present on both sides. It does not participate in the proton transfer; it simply "spectates." Cancel the Na⁺(aq) from both sides.
-
Write the Net Ionic Equation: The remaining species constitute the net ionic equation. CH₃COO⁻(aq) + H₂O(l) ⇌ CH₃COOH(aq) + OH⁻(aq)
This final equation is the definitive, simplified representation of the hydrolysis of the acetate ion. It explicitly shows the consumption of water and acetate ion and the production of acetic acid and hydroxide ion. The presence of OH⁻ is what makes the solution basic.
The Equilibrium Constant for Hydrolysis (Kb)
The extent of hydrolysis is governed by an equilibrium constant, specifically the base dissociation constant (Kb) for the acetate ion. For the net ionic equation: CH₃COO⁻(aq) + H₂O(l) ⇌ CH₃COOH(aq) + OH⁻(aq)
The equilibrium constant expression is: Kb = [CH₃COOH][OH⁻] / [CH₃COO⁻]
This Kb value is not typically tabulated directly but is intrinsically linked to the acid dissociation constant (Ka) of its conjugate acid, acetic acid, through the ion product of water (Kw): Ka(CH₃COOH) × Kb(CH₃COO⁻) = Kw = 1.0 × 10⁻¹⁴ at 25°C
Given that the Ka for acetic acid is approximately 1.8 × 10⁻⁵, we can calculate: Kb = Kw / Ka = (1.0 ×
10⁻¹⁴) / (1.8 × 10⁻⁵) ≈ 5.56 × 10⁻¹⁰.
This very small Kb value confirms that the acetate ion is a weak base. Consequently, the hydrolysis reaction lies heavily to the left, meaning only a tiny fraction of acetate ions react with water. Nevertheless, the production of even a small amount of hydroxide ion (OH⁻) is sufficient to shift the pH above 7.
To predict the pH of a sodium acetate solution, one can use the Kb expression. For a 0.10 M NaC₂H₃O₂ solution, the initial [CH₃COO⁻] ≈ 0.10 M. Assuming x M of acetate hydrolyzes: Kb = [CH₃COOH][OH⁻] / [CH₃COO⁻] ≈ (x)(x) / (0.10 - x) ≈ x² / 0.10 Solving for x (which equals [OH⁻]): x² = Kb × 0.10 = (5.56 × 10⁻¹⁰) × 0.10 = 5.56 × 10⁻¹¹ x = [OH⁻] ≈ 7.46 × 10⁻⁶ M pOH = -log(7.46 × 10⁻⁶) ≈ 5.13 pH = 14.00 - pOH ≈ 8.87
This calculated basic pH aligns perfectly with the net ionic equation’s prediction: the consumption of water by the acetate ion generates hydroxide ions.
Conclusion
The hydrolysis of sodium acetate elegantly demonstrates the fundamental principle that the conjugate base of a weak acid will react with water to produce a basic solution. The process is concisely captured by the net ionic equation CH₃COO⁻(aq) + H₂O(l) ⇌ CH₃COOH(aq) + OH⁻(aq), which isolates the essential proton-transfer event. The equilibrium constant for this reaction, Kb, is directly derived from the Ka of the conjugate acid (acetic acid) and the autoionization constant of water (Kw), illustrating the intimate link between acid and base strength. While the extent of hydrolysis is limited by the small Kb of acetate, the resulting hydroxide concentration is significant enough to elevate the pH measurably above 7. Thus, any soluble salt containing the conjugate base of a weak acid—such as sodium carbonate, sodium bicarbonate, or sodium phosphate—will exhibit basic aqueous behavior through this same hydrolytic mechanism.
This principle extends universally to all soluble salts derived from a weak acid and a strong base. The anion, as the conjugate base of that weak acid, will invariably undergo hydrolysis to some degree, generating hydroxide ions and producing a basic solution. The magnitude of the pH increase depends directly on the strength of the parent weak acid—the weaker the acid (smaller Ka), the stronger its conjugate base (larger Kb), and the more pronounced the basicity of the salt solution. Conversely, salts formed from a weak base and a strong acid (e.g., ammonium chloride) yield acidic solutions due to the hydrolysis of the cations, following an analogous proton-transfer mechanism with water. Salts of strong acids and strong bases, like NaCl, are neutral because neither ion reacts appreciably with water. Thus, by identifying the ions in a salt and referencing the appropriate Ka or Kb values, one can reliably predict the aqueous pH without experimental measurement. This predictive framework is a cornerstone of aqueous geochemistry, environmental science, and biochemistry, where the hydrolysis of species like carbonate, phosphate, and amino acid ions critically influences the pH of natural waters, biological fluids, and reaction media. In summary, salt hydrolysis is not an anomaly but a direct, quantifiable consequence of the equilibrium between conjugate acid-base pairs and water, perfectly illustrating the interconnected and self-regulating nature of acid-base chemistry in aqueous systems.