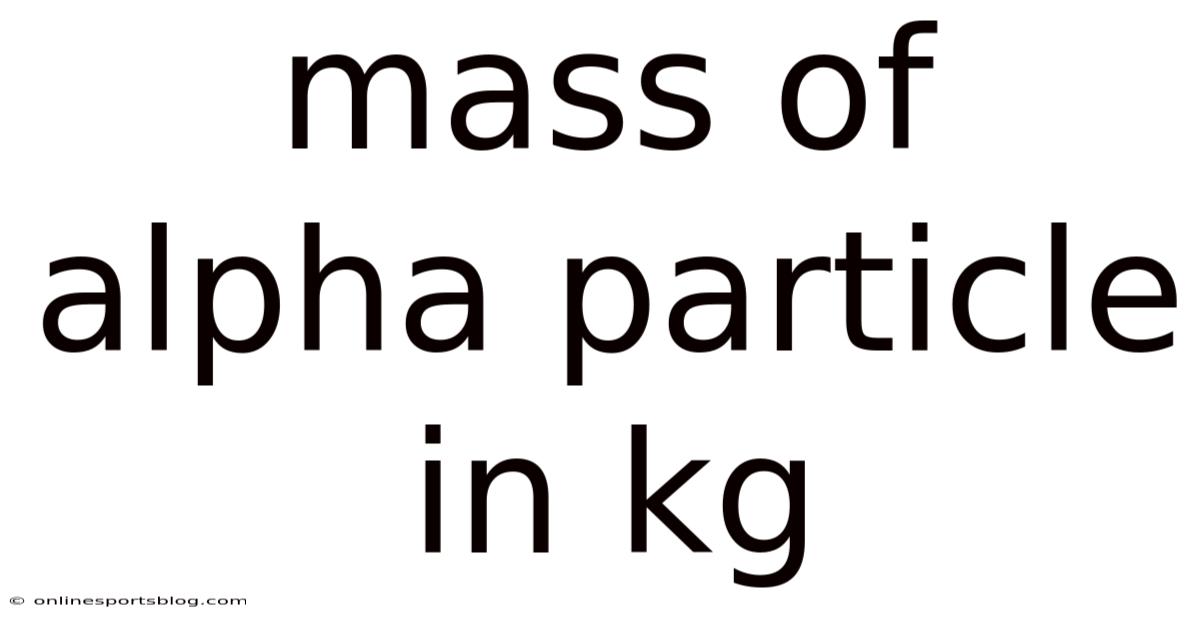Mass of Alpha Particle in kg: A Complete Guide to Understanding This Fundamental Nuclear Property
The mass of alpha particle in kg is one of the most fundamental measurements in nuclear physics, playing a crucial role in understanding radioactive decay, particle interactions, and the structure of matter at the atomic level. An alpha particle is essentially the nucleus of a helium-4 atom, consisting of two protons and two neutrons bound together. This article explores the precise mass of alpha particles, how this value is determined, and why it matters in various scientific and practical applications.
What Is an Alpha Particle?
An alpha particle is a type of ionizing radiation emitted during certain types of radioactive decay, particularly alpha decay. Unlike beta particles (which are electrons) or gamma rays (which are electromagnetic radiation), alpha particles are relatively large and heavy subatomic particles. They carry a positive charge of +2, meaning they have lost both electrons from a helium atom, leaving only the nucleus intact No workaround needed..
The composition of an alpha particle is remarkably simple yet significant:
- 2 protons: Each proton contributes approximately 1.007276 atomic mass units
- 2 neutrons: Each neutron contributes approximately 1.008665 atomic mass units
- Total nucleons: 4 (giving it a mass number of 4)
Worth pausing on this one But it adds up..
This combination makes the alpha particle one of the most tightly bound nuclear configurations in nature, which explains why alpha decay releases such significant energy when it occurs.
The Precise Mass of Alpha Particle in kg
The mass of an alpha particle in kg is approximately 6.On top of that, 644657 × 10⁻²⁷ kilograms. This value represents one of the most precisely measured quantities in physics, with multiple experimental methods confirming its accuracy to remarkable precision.
For reference purposes, here are the key numerical values:
- Mass in kilograms: 6.644657 × 10⁻²⁷ kg
- Mass in atomic mass units (u): 4.001506179127 u
- Mass in MeV/c²: 3727.379378 MeV/c²
The atomic mass unit, defined as 1/12 of the mass of a carbon-12 atom, provides a convenient reference for expressing particle masses. One atomic mass unit equals approximately 1.660539 × 10⁻²⁷ kg, which explains why the alpha particle mass, at about 4 atomic mass units, corresponds to roughly 6.64 × 10⁻²⁷ kg.
How Scientists Determine the Mass of Alpha Particle
The precise measurement of alpha particle mass involves several sophisticated techniques that physicists have developed over decades of research. Understanding these methods reveals the incredible precision achievable in modern physics.
Mass Spectrometry
Mass spectrometry represents one of the primary methods for determining particle masses. On top of that, in this technique, charged particles are passed through magnetic and electric fields, causing them to follow curved paths. The radius of curvature depends on the particle's mass-to-charge ratio. By carefully measuring these trajectories, scientists can calculate the mass of alpha particles with extraordinary precision.
Nuclear Reaction Analysis
Scientists can also determine alpha particle mass through nuclear reactions. Think about it: for example, when certain heavy nuclei undergo alpha decay, the energy of the emitted alpha particle can be precisely measured using semiconductor detectors or magnetic spectrometers. This energy, combined with knowledge of the parent and daughter nuclei masses, allows physicists to calculate the alpha particle mass indirectly but with high accuracy.
Easier said than done, but still worth knowing.
Comparison with Helium-4 Ion
Since an alpha particle is identical to a doubly-ionized helium-4 atom (He²⁺), researchers can also study helium ions in various charge states to verify alpha particle properties. The agreement between different measurement techniques provides confidence in the accepted value Still holds up..
Why the Mass of Alpha Particle Matters
The mass of alpha particles is not merely an academic curiosity—it has profound implications across multiple domains of science and technology.
Understanding Radioactive Decay
In alpha decay, a heavy radioactive nucleus (such as uranium-238 or radium-226) spontaneously emits an alpha particle, transforming into a different element. The energy released in this process, known as the Q-value, depends directly on the mass difference between the parent nucleus, daughter nucleus, and the alpha particle. Without knowing the precise alpha particle mass, scientists could not accurately calculate decay energies or predict which isotopes are stable against alpha emission Not complicated — just consistent..
Medical Applications
Alpha particles have significant applications in medicine, particularly in cancer treatment. Targeted alpha therapy uses alpha-emitting radioisotopes to deliver highly localized radiation doses to cancer cells while minimizing damage to surrounding healthy tissue. Understanding alpha particle mass helps researchers design more effective treatment protocols and calculate appropriate dosages.
Industrial and Commercial Uses
Alpha particles from radioactive sources have practical applications in smoke detectors (where americium-241 emits alpha particles that ionize air molecules), thickness gauges for manufacturing, and as neutron sources when combined with certain materials. Each application requires accurate knowledge of alpha particle properties Which is the point..
Fundamental Physics Research
In particle physics and cosmology, the alpha particle serves as a benchmark for understanding nuclear forces. Its binding energy—the energy required to separate it into individual protons and neutrons—provides crucial data for testing theoretical models of nuclear structure. The mass of the alpha particle also relates to fundamental constants and helps verify the consistency of physical theories And that's really what it comes down to..
The Alpha Particle in Context: Comparing Masses
To appreciate the significance of the alpha particle mass, it helps to compare it with other particles:
| Particle | Mass (kg) | Mass (u) |
|---|---|---|
| Alpha particle | 6.Still, 644 × 10⁻²⁷ | 4. 0015 |
| Proton | 1.672 × 10⁻²⁷ | 1.0073 |
| Neutron | 1.675 × 10⁻²⁷ | 1.0087 |
| Electron | 9.109 × 10⁻³¹ | 0. |
The official docs gloss over this. That's a mistake.
This comparison reveals that an alpha particle is approximately 4 times heavier than a proton and nearly 7,300 times heavier than an electron. This significant mass means alpha particles interact strongly with matter, losing energy quickly and traveling relatively short distances compared to other forms of radiation Most people skip this — try not to..
Frequently Asked Questions
How was the mass of alpha particle first measured?
Early measurements relied on cloud chambers and emulsion techniques, where scientists could observe alpha particle tracks and deduce properties from their behavior in magnetic fields. Modern measurements use much more sophisticated equipment, achieving precision to many decimal places.
Is the alpha particle mass constant?
Yes, the rest mass of an alpha particle is a fundamental constant of nature. Which means it does not change regardless of the radioactive source or the energy of the alpha particle. Even so, when alpha particles move at high speeds, their relativistic mass increases slightly according to Einstein's theory of relativity.
Why do alpha particles have exactly 4 atomic mass units?
The alpha particle contains 2 protons and 2 neutrons. Since protons and neutrons each have approximately 1 atomic mass unit, the total comes to about 4 u. The slight difference from exactly 4 u results from the binding energy that holds the alpha particle together—a manifestation of Einstein's E=mc² relationship Worth keeping that in mind..
Counterintuitive, but true.
Can alpha particles be weighed directly?
Not in the conventional sense. Scientists measure alpha particle mass indirectly through its effects on other particles and fields. The incredibly small scale of these particles makes direct weighing impossible with current technology.
Conclusion
The mass of alpha particle in kg—approximately 6.644657 × 10⁻²⁷ kg—represents one of the most precisely known quantities in all of physics. This value, equivalent to about 4 atomic mass units, reflects the combined mass of two protons and two neutrons bound together in the most stable nuclear configuration possible Practical, not theoretical..
Honestly, this part trips people up more than it should.
Understanding this fundamental property enables scientists to accurately describe radioactive decay processes, develop medical treatments, create practical technologies, and explore the fundamental nature of nuclear forces. The alpha particle, despite its simplicity, continues to serve as a cornerstone of nuclear physics research and applications.
And yeah — that's actually more nuanced than it sounds.
Whether you are a student, researcher, or simply curious about the building blocks of matter, the alpha particle mass provides a fascinating window into the quantum world where the laws of physics operate on scales far beyond our everyday experience.