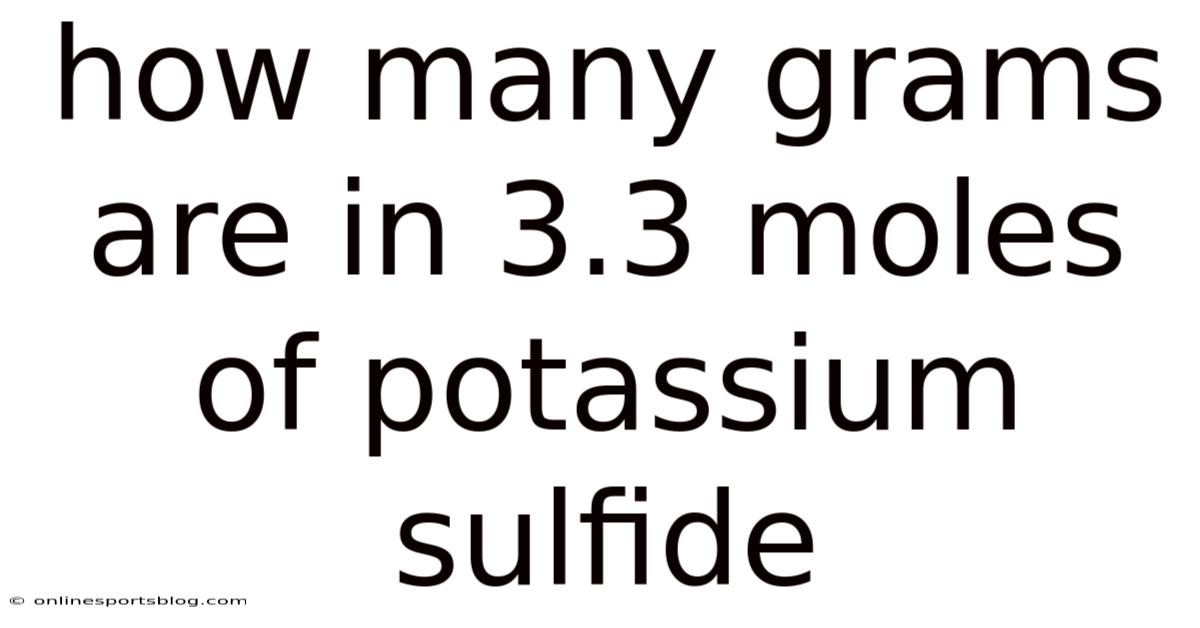The fundamental concept underlying chemistry often remains obscured despite its pervasive influence across scientific disciplines, yet understanding how to translate abstract principles into tangible reality remains a cornerstone of scientific literacy. Even so, this foundational skill thus serves as both a prerequisite and a testament to the discipline’s demands, reinforcing the importance of diligence in handling numerical transformations that directly impact experimental success. For those seeking to bridge the gap between theoretical knowledge and practical application, the task of converting moles into grams serves as a critical gateway. Consider this: in this context, the act of transforming moles into grams transcends its simplicity, becoming a microcosm of scientific rigor and precision that underpins countless disciplines. It is within this domain that the true mastery of chemistry is cultivated, where minor miscalculations can cascade into significant errors, underscoring the necessity for careful execution. That's why the challenge lies not only in the arithmetic itself but in maintaining the integrity of the process throughout the entire workflow, ensuring that each step aligns smoothly with the preceding one. This process, though seemingly straightforward at first glance, demands meticulous attention to detail and a deep appreciation for the interplay between atomic structure, molecular composition, and macroscopic measurements. Here's the thing — the process itself becomes a test of one’s ability to synthesize information, apply mathematical principles, and contextualize data within broader scientific frameworks. That said, such conversions are not merely numerical exercises; they represent the very essence of quantitative reasoning, where precision and accuracy intertwine to ensure consistency in outcomes. Whether one is a student grappling with homework or a professional navigating lab protocols, grasping the significance of converting moles into grams equips them with the foundational tools necessary to conduct experiments accurately, interpret results effectively, and communicate findings persuasively. Such attention to detail ensures that the final result not only reflects the input data but also upholds the credibility of the entire endeavor, making the conversion a central moment in any scientific investigation.
Molar mass calculations form the bedrock of such conversions, acting as the linchpin that connects atomic-scale entities to observable quantities. In the case of potassium sulfide (K₂S), the process begins with identifying its constituent elements and their respective atomic weights, which are fundamental to determining the total mass required. Potassium (K) has an atomic mass of approximately 39.1 g/mol, while sulfur (S) stands at 32.07 g/mol. Think about it: given the formula K₂S, which contains two potassium atoms paired with one sulfur atom, the calculation unfolds by multiplying the molar mass of potassium by its subscript and adding it to the sulfur’s molar mass. This step requires precise attention to the stoichiometric ratios embedded within the chemical formula, ensuring that no element is overlooked or misattributed. The resulting sum provides the precise molar mass of potassium sulfide, a value that serves as the numerical foundation for converting moles into grams. Worth adding: understanding this process demands not only familiarity with atomic weights but also an ability to manipulate numerical relationships effectively. To give you an idea, recognizing that each potassium atom contributes twice its atomic weight when paired with sulfur necessitates careful consideration of the stoichiometry involved. Such nuances are critical because even minor errors in calculation can propagate through subsequent steps, leading to cascading mistakes in experimental outcomes. On top of that, the process thus becomes a exercise in both mathematical acumen and conceptual clarity, where the correct application of principles ensures the validity of the final result. That said, this foundational knowledge empowers practitioners to approach similar calculations with confidence, reinforcing the reliability of their work. What's more, the process itself is a pedagogical tool, allowing learners to internalize the logic behind conversions and apply it across diverse contexts. Also, by mastering this aspect, individuals not only enhance their technical proficiency but also develop a systematic mindset that applies universally, whether in laboratory settings or academic pursuits. The meticulous nature of this task also highlights the importance of consistency, as repeated practice solidifies understanding and reduces reliance on rote memorization Worth keeping that in mind..
This is the bit that actually matters in practice.
underpinned by a deep appreciation for the interconnectedness of chemistry’s fundamental principles. Worth adding: for instance, in drug development, precise molar calculations ensure accurate dosing, where even a miscalculation could render a compound ineffective or hazardous. This skill transcends the confines of the classroom, serving as a cornerstone for innovation in fields ranging from pharmaceuticals to materials science. Similarly, in environmental science, converting moles to grams enables researchers to quantify pollutants in air or water samples, guiding remediation efforts with quantifiable data Simple, but easy to overlook..
The meticulousness required in these calculations also cultivates a mindset of rigor that permeates all scientific inquiry. Consider the synthesis of novel materials: engineers rely on stoichiometric precision to tailor properties like conductivity or reactivity, knowing that deviations can compromise the integrity of the final product. Such applications underscore the real-world stakes of mastering molar conversions, where theoretical knowledge directly impacts technological advancement and public safety That's the part that actually makes a difference..
Worth adding, the process of converting moles to grams fosters critical thinking by challenging individuals to contextualize abstract concepts within tangible scenarios. Students who grasp the rationale behind each step—rather than merely memorizing formulas—are better equipped to troubleshoot experimental anomalies or adapt methodologies to novel problems. This adaptability is invaluable in research, where unexpected variables often demand on-the-spot adjustments grounded in a solid theoretical foundation Simple, but easy to overlook..
All in all, the ability to convert moles to grams is far more than a routine exercise; it is a gateway to understanding the quantitative language of chemistry. By bridging the gap between atomic-scale phenomena and macroscopic measurements, this skill empowers scientists to innovate responsibly, communicate findings with clarity, and contribute to a discipline that thrives on precision. As chemistry continues to address global challenges—from sustainable energy to medical
diagnostics – the proficiency in converting moles to grams will remain an indispensable tool. It's a fundamental building block, firmly establishing a foundation for complex chemical analysis and driving progress across a vast spectrum of scientific endeavors. Because of that, the seemingly simple conversion, when approached with thoughtful application, reveals the profound power of quantitative thinking and its crucial role in shaping a better future. It's not just about numbers; it's about understanding the underlying principles and using them to solve real-world problems with confidence and accuracy Which is the point..
diagnostics – the proficiency in converting moles to grams will remain an indispensable tool. Practically speaking, it's a fundamental building block, firmly establishing a foundation for complex chemical analysis and driving progress across a vast spectrum of scientific endeavors. The seemingly simple conversion, when approached with thoughtful application, reveals the profound power of quantitative thinking and its crucial role in shaping a better future. It's not just about numbers; it's about understanding the underlying principles and using them to solve real-world problems with confidence and accuracy.
Beyond the immediate applications in research and industry, the skill of converting moles to grams fosters a deeper appreciation for the interconnectedness of scientific concepts. Consider this: it encourages a holistic view, where microscopic interactions translate into observable, measurable effects. This perspective is vital for navigating the complexities of modern science, particularly in fields like biochemistry and pharmacology, where understanding drug metabolism and bioavailability hinges on precise quantitative assessments.
To build on this, the ability to perform this conversion enhances data interpretation skills. Plus, in experimental settings, raw data often needs to be transformed and analyzed to reveal meaningful insights. Molar conversions are frequently a necessary step in this process, allowing researchers to compare results across different experiments or to correlate chemical properties with observed phenomena. This analytical capability is essential for drawing valid conclusions and making informed decisions based on scientific evidence That's the whole idea..
The bottom line: mastering molar conversions is an investment in scientific literacy and critical thinking. It’s a skill that transcends the laboratory, contributing to a greater understanding of the chemical processes that underpin our everyday lives, from the food we eat to the materials we use. It empowers individuals to not only understand the language of chemistry but also to engage with the world around them in a more informed and discerning manner. And as scientific challenges become increasingly complex, the foundational ability to convert moles to grams will continue to be a cornerstone of innovation and progress.