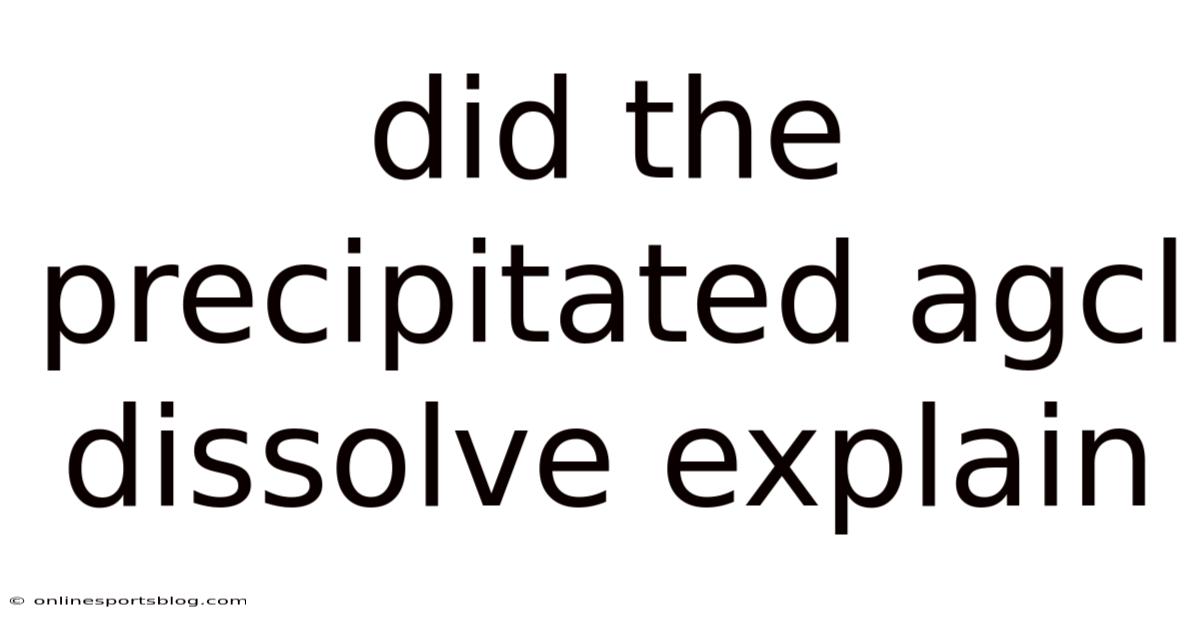When silver chloride (AgCl) is formed in a solution, it often appears as a white precipitate. This phenomenon occurs because AgCl is highly insoluble in water. Even so, the question of whether the precipitated AgCl can dissolve requires a deeper understanding of solubility equilibria and chemical reactions And that's really what it comes down to. Practical, not theoretical..
Silver chloride is a sparingly soluble salt, and its solubility in water is governed by the solubility product constant (Ksp). At room temperature, the Ksp of AgCl is approximately 1.8 x 10^-10, indicating that very little AgCl can dissolve in water Took long enough..
AgCl(s) ⇌ Ag⁺(aq) + Cl⁻(aq)
Despite its low solubility, AgCl can dissolve under certain conditions. One common method to dissolve AgCl is by adding ammonia (NH₃) to the solution. Plus, ammonia forms a complex ion with silver, known as the diamminesilver(I) complex [Ag(NH₃)₂]⁺. This reaction reduces the concentration of free Ag⁺ ions in the solution, shifting the equilibrium to the right and causing more AgCl to dissolve.
This is the bit that actually matters in practice.
AgCl(s) + 2NH₃(aq) ⇌ [Ag(NH₃)₂]⁺(aq) + Cl⁻(aq)
Another way to dissolve AgCl is by adding a solution with a high concentration of chloride ions, such as concentrated hydrochloric acid (HCl). This process involves the formation of the tetrachloroargentate(I) complex ion, [AgCl₄]³⁻. The reaction is as follows:
AgCl(s) + Cl⁻(aq) ⇌ [AgCl₂]⁻(aq)
In both cases, the dissolution of AgCl is driven by the formation of complex ions, which effectively remove Ag⁺ ions from the equilibrium and allow more AgCl to dissolve.
Worth pointing out that the dissolution of AgCl is not a simple process and depends on the specific conditions of the solution. In real terms, factors such as temperature, concentration of reactants, and the presence of other ions can influence the extent to which AgCl dissolves. Here's one way to look at it: increasing the temperature generally increases the solubility of most salts, including AgCl, although the effect may be minimal due to its low solubility.
To keep it short, while AgCl is highly insoluble in water, it can dissolve under specific conditions, such as the addition of ammonia or concentrated chloride solutions. The dissolution process involves the formation of complex ions, which shift the solubility equilibrium and allow more AgCl to dissolve. Understanding these principles is crucial for predicting and controlling the behavior of AgCl in various chemical processes.