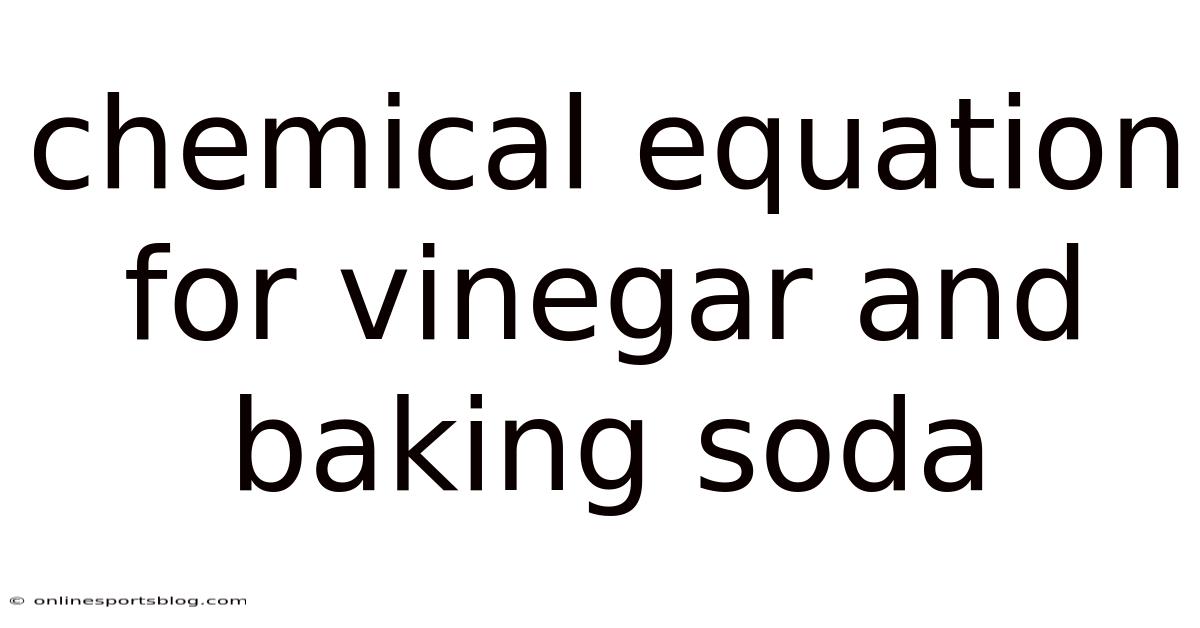Introduction
The chemical equation for vinegar and baking soda is a classic example of an acid‑base reaction that produces carbon dioxide gas, water, and a salt. This reaction is not only a staple in school laboratories but also the scientific principle behind many everyday phenomena, from leavening baked goods to cleaning surfaces. In this article we will explore the balanced equation, the steps to derive it, the underlying science, practical uses, and answer common questions. Understanding the chemical equation for vinegar and baking soda equips readers with a tangible glimpse into how simple household ingredients can generate noticeable chemical change.
This changes depending on context. Keep that in mind.
Steps
To write the balanced equation, follow these systematic steps:
- Identify the reactants – Vinegar is an aqueous solution of acetic acid (CH₃COOH), and baking soda is sodium bicarbonate (NaHCO₃). 2. Write the unbalanced molecular equation – Combine the two reactants and indicate the products that are typically observed:
[ \text{CH}_3\text{COOH (aq)} + \text{NaHCO}_3 \text{ (s)} \rightarrow \text{CO}_2 \text{ (g)} + \text{H}_2\text{O (l)} + \text{
CH₃COONa (aq).
This completes the unbalanced molecular equation:
[ \text{CH}_3\text{COOH (aq)} + \text{NaHCO}_3 \text{ (s)} \rightarrow \text{CO}_2 \text{ (g)} + \text{H}_2\text{O (l)} + \text{CH}_3\text{COONa (aq)} ]
Balancing the Equation
To verify that the equation is balanced, count each type of atom on both sides of the arrow:
| Atom | Reactant side | Product side |
|---|---|---|
| C | 2 (from CH₃COOH) + 1 (from NaHCO₃) = 3 | 1 (in CO₂) + 2 (in CH₃COONa) = 3 |
| H | 4 (from CH₃COOH) + 1 (from NaHCO₃) = 5 | 2 (in H₂O) + 3 (in CH₃COONa) = 5 |
| O | 2 (from CH₃COOH) + 3 (from NaHCO₃) = 5 | 2 (in CO₂) + 1 (in H₂O) + 2 (in CH₃COONa) = 5 |
| Na | 1 (from NaHCO₃) | 1 (in CH₃COONa) |
All atom counts match, so the equation is already balanced with a 1 : 1 stoichiometric ratio.
The net ionic equation, which removes the spectator Na⁺ ion, is:
[ \text{CH}_3\text{COOH (aq)} + \text{HCO}_3^- \text{ (aq)} \rightarrow \text{CO}_2 \text{ (g)} + \text{H}_2\text{O (l)} + \text{CH}_3\text{COO}^- \text{ (aq)} ]
Underlying Science
Acid‑base classification – According to the Brønsted‑Lowry definition, acetic acid (CH₃COOH) acts as a proton donor, while sodium bicarbonate (NaHCO₃) serves as a proton acceptor. The bicarbonate ion (HCO₃⁻) is the conjugate base of carbonic acid (H₂CO₃).
Reaction mechanism – The process occurs in two elementary steps:
-
Proton transfer: The acidic hydrogen of acetic acid is transferred to the bicarbonate ion, forming carbonic acid and acetate ion:
[ \text{CH}_3\text{COOH} + \text{HCO}_3^- \rightarrow \text{H}_2\text{CO}_3 + \text{CH}_3\text{COO}^- ]
-
Decomposition of carbonic acid: Carbonic acid is unstable under ambient conditions and spontaneously decomposes into carbon dioxide and water:
[ \text{H}_2\text{CO}_3 \rightarrow \text{CO}_2 \text{ (g)} + \text{H}_2\text{O (l)} ]
The overall result is the liberation of CO₂ gas, which manifests as the familiar fizzing and bubbling Still holds up..
Energetics – The reaction is exothermic, releasing approximately 50 kJ mol⁻¹ of heat. This warmth can be felt when vinegar and baking soda are mixed in a container, making the reaction useful for simple thermal demonstrations Worth keeping that in mind..
pH change – Vinegar has a pH of about 2–3 (strongly acidic). After the reaction, the solution contains sodium acetate, a salt of a weak acid and a strong base, resulting in a mildly basic pH (≈8–9). The shift from acidic to basic illustrates a complete neutralization Easy to understand, harder to ignore..
Practical Uses
- Leavening in baking – In doughs and batters, the CO₂ bubbles expand during cooking, creating a porous, fluffy texture. This is the principle behind “quick breads” such as pancakes and muffins.
- Cleaning agent – The fizzing action helps physically agitate and lift stains, while the slight alkalinity of the resulting acetate solution aids in dissolving greasy residues. Many homemade cleaning recipes combine vinegar and baking soda for this reason.
- Educational demonstrations – The classic “volcano” model uses the vigorous evolution of CO₂ to simulate an eruption, providing a vivid illustration of gas production and acid‑base chemistry.
- Mild deodorizer – The basic acetate ions can neutralize acidic odors, while the released CO₂ helps disperse volatile compounds.
Common Questions
Is the reaction reversible?
Under ordinary conditions, the escape of CO₂ gas drives the reaction forward. If the CO₂ were trapped and allowed to dissolve back into solution, the reverse reaction (formation of carbonic acid) could occur, but the overall equilibrium lies far to the right Turns out it matters..
Can other acids be substituted for vinegar?
Yes. Lemon juice (citric acid), cream of tartar (tartaric acid), or even hydrochloric acid will react with baking soda to produce CO₂, water, and the corresponding salt. The rate and intensity of fizzing may vary with the acid’s strength and concentration.
What is the theoretical yield of CO₂?
One mole of each reactant yields one mole of CO₂. For practical purposes, a teaspoon (≈5 g) of baking soda mixed with an equivalent amount of vinegar (≈5 ml of 5 % acetic acid) generates roughly 1 L of CO₂ gas at room temperature, though actual yield depends on temperature, pressure, and exact concentrations Surprisingly effective..
Is the reaction safe?
The products (CO₂, water, and sodium acetate) are non‑toxic. Even so, the rapid gas evolution can cause pressure buildup in sealed containers, leading to possible splattering or bursting. Always conduct the reaction in an open vessel or a loosely capped container.
Why does the mixture feel warm?
The exothermic nature of the acid‑base neutralization releases heat. The temperature increase is typically modest (a few degrees Celsius) but can be noticeable Worth knowing..
Conclusion
The chemical equation for vinegar and baking soda encapsulates a fundamental acid‑base neutralization that is both simple enough for classroom demonstration and versatile enough for everyday applications. The visible fizzing, heat release, and pH change provide tangible evidence of chemical transformation, making this reaction an enduring tool for teaching, cleaning, cooking, and scientific outreach. On top of that, by transferring a proton from acetic acid to bicarbonate, the reaction produces carbon dioxide gas, water, and sodium acetate—a salt that shifts the pH from acidic to mildly basic. Understanding this classic reaction not only demystifies a common household phenomenon but also illustrates core concepts in stoichiometry, thermodynamics, and reaction mechanics that form the bedrock of chemistry.
Practical Tips for Getting the Most Out of the Reaction
| Goal | Recommended Ratio | Mixing Technique | Additional Add‑ins |
|---|---|---|---|
| Maximum fizz | 1 part baking soda : 1 part 5 % vinegar (by weight) | Add the dry powder slowly to the liquid while stirring gently; a rapid dump will cause premature foaming and loss of CO₂. Day to day, | A pinch of citric acid or a few drops of dish‑soap to stabilize bubbles and create a longer‑lasting foam. Day to day, |
| Steady gas flow for inflating a balloon | 2 parts baking soda : 1 part vinegar (by volume) | Dissolve the soda in a small amount of water first; then pour the vinegar into a bottle, quickly attach a balloon, and invert the mixture into the balloon. In practice, | |
| Controlled heat for a “thermal” experiment | 1 part baking soda : 2 parts concentrated vinegar (≈10 % acetic acid) | Use a heat‑resistant glass beaker; add the acid to the base rather than the reverse to avoid splatter. | A thin layer of aluminum foil beneath the beaker can reflect heat, making the temperature rise more apparent. |
Scaling the Reaction
Because the stoichiometry is 1:1, scaling is straightforward: double both masses, double the volume of vinegar, and the amount of CO₂ produced will double as well. On the flip side, when moving from a kitchen‑scale experiment (grams) to a laboratory‑scale one (kilograms), several practical considerations become important:
- Gas Capture – For quantitative work, direct the evolving CO₂ through a water trap or a gas‑syringe. Using a pressure‑rated vessel (e.g., a 2 L pressure flask with a vented stopcock) prevents over‑pressurisation.
- Heat Management – The reaction’s enthalpy (≈ – 57 kJ mol⁻¹) becomes significant at larger scales; a water‑bath or cooling jacket may be required to keep the temperature within safe limits.
- Mixing Efficiency – At high solid‑to‑liquid ratios, the bicarbonate can form a slurry that slows diffusion. Mechanical stirring or a vortex mixer ensures uniform contact and consistent gas evolution.
Extending the Chemistry: From Simple Fizz to Functional Materials
While the classic vinegar‑baking‑soda reaction is most often presented as a curiosity, chemists have harnessed the underlying acid‑base neutralization in more sophisticated contexts:
- Porous Materials – The rapid generation of CO₂ within a polymerizing resin creates a “foaming” effect, yielding lightweight, insulating foams used in construction and packaging. By adjusting the acid/base pair and the resin’s cure kinetics, manufacturers can tailor pore size and density.
- Carbon Capture Demonstrations – Sodium bicarbonate is a convenient solid sorbent for CO₂. In a reverse setup, bubbling CO₂ through an aqueous sodium acetate solution reforms sodium bicarbonate, illustrating the cyclical nature of carbon capture and release.
- Microreactor Propulsion – In micro‑robotics, the localized production of CO₂ from acid‑base pairs powers tiny “jet” thrusters, enabling controlled movement without external pumps. The reaction’s simplicity and low toxicity make it attractive for biomedical devices that must operate in aqueous environments.
Safety Checklist (For the Enthusiast)
- Ventilation – Although CO₂ is non‑toxic at low concentrations, accumulation in a confined space can displace oxygen. Perform the reaction in a well‑ventilated area.
- Eye Protection – Splashing vinegar can cause mild irritation; safety goggles are recommended, especially when scaling up.
- Container Choice – Use glass or sturdy plastic; avoid sealed metal containers, as CO₂ pressure can cause rupture.
- Temperature Monitoring – If using concentrated acetic acid or large quantities, monitor the temperature with a thermometer; a sudden spike may indicate an exothermic runaway.
- Disposal – The resulting sodium acetate solution can be poured down the drain with plenty of water. No special hazardous‑waste procedures are required.
Final Thoughts
The vinegar‑baking‑soda reaction epitomises how a straightforward acid‑base neutralization can cascade into a suite of observable phenomena: gas evolution, temperature change, pH shift, and the formation of a useful salt. Now, by dissecting each step—from proton transfer to carbonate decomposition—students and hobbyists alike gain a concrete grasp of fundamental chemical principles. On top of that, the reaction’s versatility extends far beyond the kitchen table, informing industrial foam production, educational demonstrations of gas laws, and even emerging technologies in soft robotics and carbon management.
In essence, this humble fizz is a microcosm of chemistry itself: a balanced interplay of reactants, energy, and products that can be scaled, tweaked, and repurposed to solve real‑world problems. Appreciating its underlying mechanisms not only enriches our scientific literacy but also empowers us to innovate with the simplest of reagents—vinegar and baking soda It's one of those things that adds up. Worth knowing..