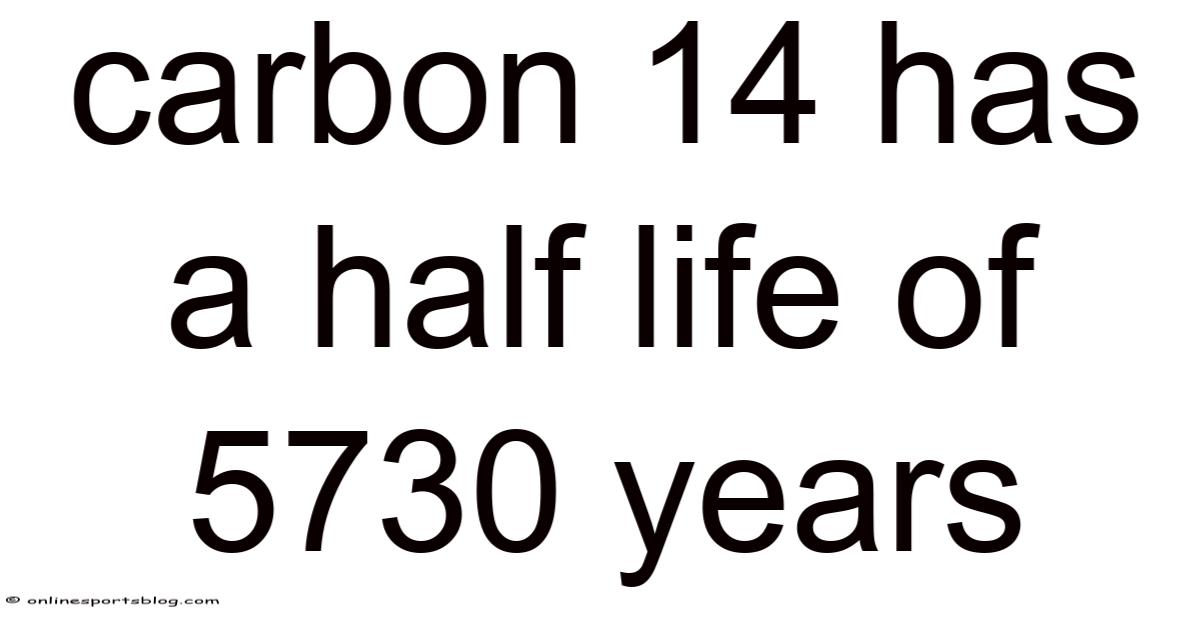Carbon 14 Has a Half Life of 5730 Years
Carbon-14, a radioactive isotope of carbon, has a half-life of 5,730 years, making it one of the most valuable tools in archaeology, geology, and environmental science. This unique property allows scientists to determine the age of once-living organisms with remarkable precision, opening a window into the past that spans tens of thousands of years. Understanding why this half-life matters and how it is used in practice reveals the elegant intersection of nuclear physics and real-world discovery.
What Is Carbon-14?
Carbon exists in nature in several forms, known as isotopes. The most common and stable forms are Carbon-12 and Carbon-13, which remain unchanged over time. Worth adding: carbon-14, however, is different. It is a radioactive isotope, meaning its atomic nucleus is unstable and will eventually decay into another element over time Small thing, real impact..
Carbon-14 contains six protons and eight neutrons, giving it an atomic mass of 14. This extra pair of neutrons makes the nucleus unstable, triggering a process called beta decay, in which a neutron converts into a proton, emitting an electron and an antineutrino in the process. The result is a stable atom of Nitrogen-14.
The key question is: how long does this transformation take? The answer lies in the concept of half-life.
Understanding Half-Life
A half-life is the time required for exactly half of a given quantity of a radioactive substance to decay into its daughter product. It is not a linear process where the material simply vanishes after a set period. Instead, it follows an exponential decay curve Simple, but easy to overlook..
This changes depending on context. Keep that in mind Simple, but easy to overlook..
To visualize this, imagine you start with 100 grams of pure Carbon-14:
- After 5,730 years, 50 grams remain.
- After 11,460 years (two half-lives), 25 grams remain.
- After 17,190 years (three half-lives), 12.5 grams remain.
- After 22,920 years (four half-lives), 6.25 grams remain.
- After 28,650 years (five half-lives), 3.125 grams remain.
Each half-life reduces the remaining amount by exactly half, but the absolute quantity lost becomes smaller with each successive interval. This predictable pattern is what makes radioactive dating so reliable.
Why the 5,730-Year Half-Life Matters
The half-life of Carbon-14 is not too short and not too long — it sits in a scientifically ideal range. On the flip side, if the half-life were only a few days or weeks, the isotope would vanish almost immediately after an organism died, leaving no measurable signal. If it were millions or billions of years, the decay would be so slow that the difference between a young sample and an old sample would be nearly impossible to detect Easy to understand, harder to ignore..
Quick note before moving on.
At 5,730 years, Carbon-14 provides a practical dating window of approximately 50,000 to 60,000 years. Beyond this range, the remaining Carbon-14 in a sample drops to levels too small to measure accurately with standard equipment. Within this window, however, the isotope serves as a remarkably precise chronological clock Worth keeping that in mind..
How Carbon-14 Is Formed
Carbon-14 is not a product of life on Earth. Even so, high-energy particles from space, primarily protons, constantly bombard the Earth's upper atmosphere. Think about it: it is created high in the atmosphere through a process driven by cosmic radiation. When these particles collide with atoms of Nitrogen-14, they knock a neutron out of the nitrogen nucleus and replace it with a new one, effectively converting Nitrogen-14 into Carbon-14.
Once formed, Carbon-14 rapidly oxidizes to create carbon dioxide (CO₂). This CO₂ mixes with the atmosphere and is absorbed by plants during photosynthesis. But animals then consume the plants, and the Carbon-14 enters the food chain. As long as an organism is alive, it continuously exchanges carbon with its environment, maintaining a steady ratio of Carbon-14 to Carbon-12 that mirrors the atmosphere Not complicated — just consistent..
The moment an organism dies, however, this exchange stops. Because of that, the Carbon-14 trapped in its tissues begins to decay at a fixed rate, while the stable Carbon-12 remains unchanged. By measuring the ratio of Carbon-14 to Carbon-12 in a sample, scientists can calculate how long ago the organism died And that's really what it comes down to..
How Carbon-14 Dating Works in Practice
The process of radiocarbon dating involves several critical steps:
- Sample Collection: Scientists carefully extract organic material from the specimen being studied. Suitable samples include wood, bone, shell, charcoal, seeds, and even hair.
- Preparation: The sample is cleaned to remove contaminants such as rootlets, soil, or modern carbon that could skew the results.
- Conversion: The organic carbon in the sample is converted into a measurable form, typically graphite or carbon dioxide gas.
- Measurement: The ratio of Carbon-14 to Carbon-12 is measured using one of several techniques:
- Accelerator Mass Spectrometry (AMS): This modern method directly counts individual Carbon-14 atoms, requiring only tiny sample sizes.
- Liquid Scintillation Counting: This older technique measures the beta particles emitted during decay.
- Gas Proportional Counting: This method counts beta particles in a gas-filled chamber.
- Calibration: Raw radiocarbon dates are calibrated against known records of atmospheric Carbon-14 levels, primarily derived from tree rings (dendrochronology) and coral records, to produce a more accurate calendar age.
Applications of Carbon-14 Dating
The 5,730-year half-life of Carbon-14 has made it indispensable across multiple fields:
- Archaeology: Dating artifacts such as wooden tools, textiles, and human remains to establish timelines of ancient civilizations.
- Geology: Studying sediment layers and fossilized organic material to reconstruct past environments.
- Climate Science: Analyzing ice cores, tree rings, and ocean sediments to understand historical climate patterns.
- Forensics: Determining the age of unidentified human remains or detecting fraud in the art and antiquities market.
- Oceanography: Tracking ocean circulation patterns by measuring Carbon-14 levels in deep-sea corals and marine organisms.
Limitations of Carbon-14 Dating
Despite its power, radiocarbon dating has important limitations that researchers must account for:
- Age Range: The method is effective only for samples up to about 50,000 to 60,000 years old. Beyond this, the remaining Carbon-14 is too faint to detect reliably.
- Contamination: Even small amounts of modern carbon can dramatically skew results, making careful sample handling essential.
- Reservoir Effects: Variations in Carbon-
Reservoir Effects (Continued)
Marine organisms, freshwater fish, and even some terrestrial plants can incorporate “old” carbon that has been out of exchange with the atmosphere for centuries or millennia. This reservoir effect causes their apparent radiocarbon ages to be older than their true calendar ages. On the flip side, to correct for this, researchers apply region‑specific offset values derived from known-age samples (e. Which means g. , historically dated shells or fish otoliths). In some cases, especially for deep‑sea corals, the offset can be several hundred years, and failure to account for it can introduce significant error into climate reconstructions It's one of those things that adds up..
Calibration Curve Uncertainties
The calibration curve (IntCal) is built from a mosaic of tree‑ring sequences, varved lake sediments, and corals. In practice, while it provides an excellent match for most time periods, there are intervals—particularly around 12,000–8,000 BP (the early Holocene) and 2,500–1,500 BP (the “Hallstatt plateau”)—where the curve flattens. That said, in these zones, a range of radiocarbon ages corresponds to a relatively narrow calendar span, making precise dating difficult. Think about it: researchers often complement C‑14 results with other dating methods (e. On the flip side, g. , dendrochronology, optically stimulated luminescence) to resolve such ambiguities Easy to understand, harder to ignore..
Sample Size and Preservation
Older samples contain fewer Carbon‑14 atoms, demanding larger amounts of material for accurate measurement. And in the case of bone or charcoal, the required mass can be several grams, which may be impractical for precious or fragile artifacts. Beyond that, diagenetic processes (chemical alteration after burial) can replace original carbon with geogenic carbon, effectively erasing the radiocarbon signal.
Recent Advances and Future Directions
1. Improved AMS Sensitivity
Modern AMS instruments now require as little as 1 mg of carbon, opening the door to dating minute specimens such as individual plant seeds, tiny insect fragments, or even single hairs. This miniaturization reduces destructive sampling, a critical benefit for museum collections Took long enough..
2. Compound‑Specific Radiocarbon Dating (CS‑RD)
Rather than measuring bulk carbon, CS‑RD isolates specific molecular compounds (e.g., amino acids, lipids) before dating. This approach can separate the original biogenic carbon from later contaminants, dramatically improving accuracy for complex matrices like sedimentary organic matter Worth keeping that in mind. Simple as that..
3. Bayesian Modeling
Software packages such as OxCal and BCal integrate radiocarbon dates with stratigraphic information, archaeological typology, and other chronological constraints. By applying Bayesian statistics, researchers generate probability distributions that reflect both the radiocarbon data and the contextual knowledge, yielding tighter chronological windows Turns out it matters..
4. Integration with Other Isotopic Systems
Combining C‑14 with other isotopic clocks—such as Uranium‑Series, Optically Stimulated Luminescence (OSL), or Cosmogenic Nuclides—provides cross‑validation and extends the temporal reach beyond the carbon‑14 limit. Multi‑proxy chronologies are becoming standard in high‑resolution paleoenvironmental studies Small thing, real impact..
5. Artificial Intelligence for Calibration
Machine‑learning algorithms are being trained on the ever‑growing database of calibration points to predict subtle, non‑linear variations in past atmospheric C‑14 production. Early results suggest that AI‑enhanced calibration could reduce the residual uncertainty of calibrated dates by up to 10 % Simple as that..
Practical Tips for Researchers New to Radiocarbon Dating
| Step | Best Practice | Why It Matters |
|---|---|---|
| Sampling | Choose the most protected part of the artifact (e. | Ensures the most up‑to‑date atmospheric curve is used. Which means |
| Statistical Reporting | Provide both the raw radiocarbon age (BP) and the calibrated calendar range with confidence intervals (68 % and 95 %). Which means | |
| Size Assessment | Verify that the sample meets the laboratory’s minimum carbon requirement. Which means | |
| Pre‑treatment | Use acid‑base‑acid (ABA) cleaning for charcoal; gelatinization for bone collagen. g.Consider this: g. | Essential for interpreting results and for future re‑analysis. |
| Documentation | Record exact provenance, stratigraphic context, and any visible contaminants. That's why | Prevents failed analyses and unnecessary sample loss. , IntCal20) and apply regional reservoir corrections if needed. , inner wood, deep bone cortex). |
| Calibration Choice | Select the appropriate IntCal version (e. | Transparent reporting allows peers to assess reliability. |
Conclusion
Carbon‑14 dating remains one of the most versatile and widely used chronometric tools in the scientific toolbox. On the flip side, its ability to translate the invisible decay of a single isotope into concrete calendar years has transformed our understanding of human history, Earth’s climate, and the dynamics of the biosphere. While the method is bounded by a finite half‑life, susceptible to contamination, and occasionally confounded by reservoir effects, continual methodological refinements—particularly in AMS sensitivity, compound‑specific analysis, and statistical modeling—are steadily expanding its precision and applicability Not complicated — just consistent. That alone is useful..
In the coming decades, the synergy between radiocarbon dating and complementary isotopic techniques, bolstered by AI‑driven calibration and ever‑larger reference databases, promises to push the frontiers of chronology further back and finer in resolution. For archaeologists, geologists, climate scientists, and forensic experts alike, mastering the nuances of C‑14 dating not only unlocks the past but also provides a dependable framework for interpreting the complex, interwoven narratives that shape our world today.